Matter is anything that occupies space and has mass. Everything around us is made of matter. You may have heard the phrase states of matter. Matter can take different forms depending on the conditions. Here on Earth matter is generally in the form of a solid, liquid or a gas.
Matter is made up of tiny particles called atoms!
How many states of matter are there?
There are actually 5 states of matter.
The three main states of matter are, solid, liquid and gas. These are the three that occur most commonly on Earth. Water is the only substance that exists in all three states at natural temperatures!
What are the 5 states of matter?
Solid
Molecules tightly packed together and don't move around much.
Particles in a solid have strong bonds between each other.
Solids have a fixed volume and shape.
The molecules in a solid can vibrate but the structure doesn't not change.
Solids cannot be compressed.
Examples of solids are: metals, wood
Liquid
Molecules more spread out and can move around and bump into each other.
Bonds between particles are weak.
Liquids have a fixed volume but can change shape. They take the shape of the container they are in.
Liquids can be slightly compressed.
Examples of liquids: water and oil
Gas
Molecules are far apart can move around freely and quickly.
Bonds between particles are very week.
The shape and volume of a gas are not fixed.
Gases can be compressed.
Examples of gases: oxygen, carbon dioxide, steam ( water vapour )
Plasma ( not the stuff that's in your blood )
Plasma is made when gases are heated to such high temperatures that electrons are ripped away from the atoms to form an ionised gas!
Almost all the visible universe is made of plasma.
We use plasma for fluorescent lights and plasma TVs but it also has lots of exciting medical uses.
Bose-Einstein Condensate
Bose-Einstein Condensate is a state of matter which forms when particles ( bosons ) are cooled to almost absolute zero.
The diagram below shows how solids, liquids and gases change state depending on temperature.
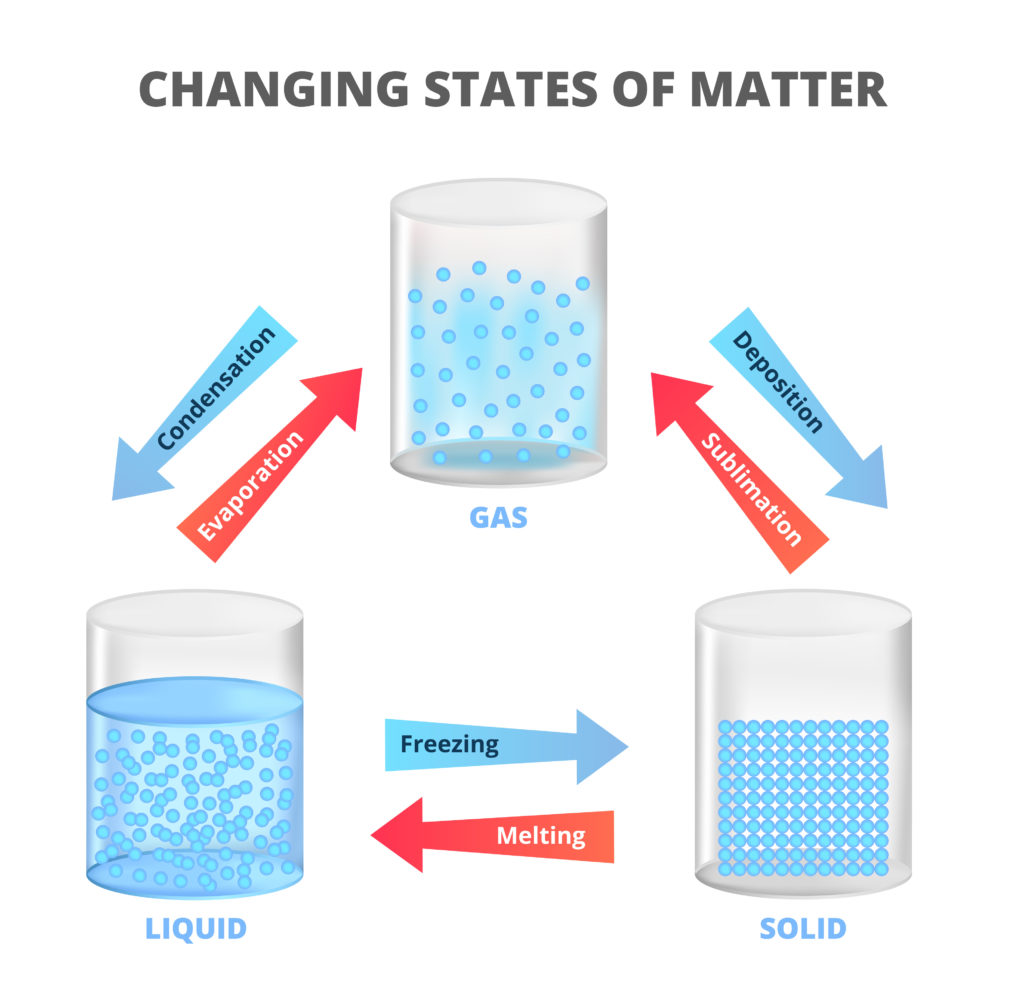
Remember - an atom is a small unit of matter and a molecule is a group of atoms bonded together.

Last Updated on July 18, 2022 by Emma Vanstone




Leave a Reply