What is a Mixture?
A mixture is a substance in which two or more substances are mixed but not chemically joined together, meaning that a chemical reaction has not taken place.
Mixtures can be easily separated and the substances in the mixture keep their original properties.
Imagine mixing skittles and full size marshmallows, the individual components (skittles and marshmallows) could easily be separated using a filter and each component of the mixture ( skittles and marshmallow ) doesn't change.
How to make a mixture
You can make your own mixtures with items from around the house.
1. Firstly try to make a mixture of toys.

2. This time use cereals or sweets.

What is a solution?
A solution is made when a solid (which we call a solute) dissolves into a liquid (that we call the solvent) One example of a solution is salt dissolved in water. The salt and water can be separated again by evaporation ( the water will evaporate if left in a hot place leaving he salt behind ).
Investigation
Aim: To test out these three mixtures to see which form solutions and which don't
- Salt and Water
- Sugar and Water
- Sand and Water
Results Table
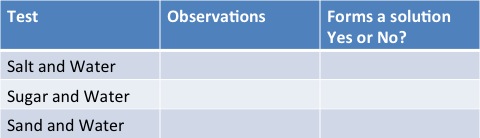
You should find that both salt and water and sugar and water dissolve and form solutions and that sand sinks to bottom!
How do you separate mixtures?
Can you separate the components out of the mixture again? Hint - to separate the sand from water you could use a sieve. This is possible as the sand is insoluble ( doesn't dissolve in water ).
Salt and sugar are soluble ( dissolve in water ) and can be separated by evaporation.
Another way to separate a mixture is by using a process called chromatography.
Challenge - how would you separate rock salt and water?
Rock salt is a mixture of salt and sand and is often spread on roads in winter to stop cars skidding.
Stage 1 - Grinding
First the rock salt should be ground using a pestle and mortar.
Stage 2- Dissolving
The ground rock salt should be dissolved in a beaker and stirred thoroughly.
Stage 3 - Filtering
The solution of water and rock salt should be passed through the filter paper where the sand ( which will not have dissolved in the water ) will collect.
Salt does dissolve in water and so will pass through the filter paper.
Stage 4 - Evaporating
To separate the salt from the water the water needs to be evaporated off, either by leaving the salty solution in the sunshine or placing under a heat source.
The salt will form as crystals - this process is called crystallisation.

Last Updated on May 24, 2021 by Emma Vanstone




Theresa says
Those are great educational play activities. I love your ideas for teaching the difference between a mixture and a solution in a meaningful way. Families can try out your mixtures and solutions and then come up with their own too. Thank you for sharing this on Artsy Play Wednesday on Capri + 3.
: 0 ) Theresa
ScienceSparks says
Thank you. I'm glad you like it. x
Khalisah Yasin says
This helps me to keep my child busy and I love it
So well done!
joy says
Simple, fun, and accurate!! Thanks from myself and my e-schooled granddaughter
😉
Ana K says
I´m a middle school student and and it has helped me understand things better