Making pH test strips is surprisingly easy and I was very impressed with how well they worked. These are a lovely alternative or additional activity to making red cabbage indicator solution and fantastic for learning about the pH scale.
How to make pH indicator paper
You'll need
Red Cabbage Indicator - made by boiling chopped red cabbage in water for 10 minutes
Vinegar/baking soda/lemon juice to test
How to make pH test strips
Dip a piece of filter paper into the red cabbage indicator and leave to dry.
Once dry, drop the solutions to test onto the paper and watch as the colour changes

pH Indicator and the pH scale
Solutions can be acidic, alkaline or neutral:
- an acidic solution is when an acid is dissolved in water
- an alkaline solution is when an alkali is dissolved in water
- solutions that are neither acidic nor alkaline are neutral.
Pure water is neutral.
What is a pH indicator?
An indicator is a substance that changes colour when added to an acidic or alkaline solution.
In a laboratory either Litmus or Universal Indicators are used. Litmus can only tell us if a substance is an acid or alkali while Universal Indicator tells us how strong the acid or alkali is.
What is Universal Indicator?
Universal Indicator shows the pH of a substance as it changed colour depending on the strength of the acid or alkali.
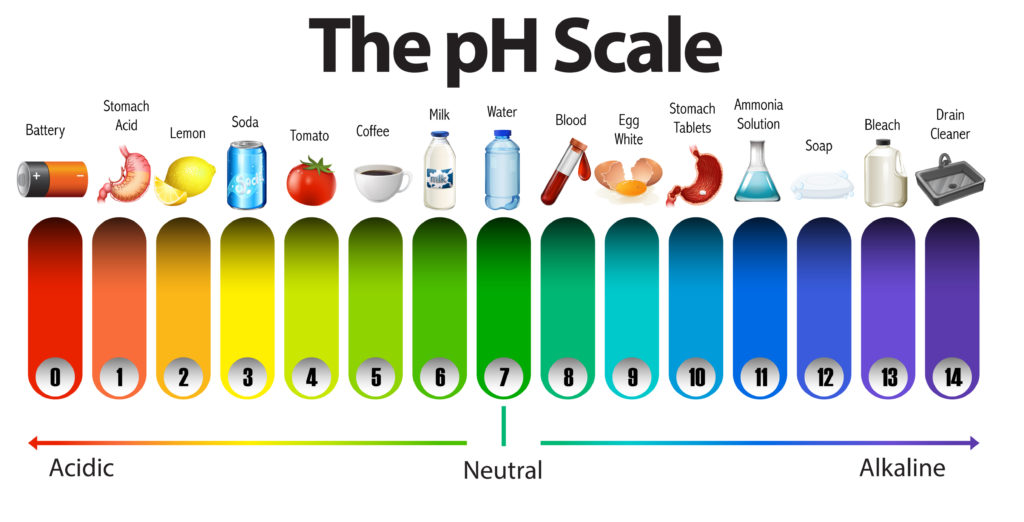
Facts about Acids
Acids have a low pH ( below 7 )
Acids turn pH paper red or orange
Acids taste sour.
Some acids can damage living cells.
Acids are neutralised by bases to make salts and water.
Acids make hydrogen when they react with some metals.
Acids have hydrogen in them! For example, Hydrocholoric Acid ( HCl )
Facts about Alkalis
Alkalis have a high pH ( over 7 ) and turn pH paper blue or purple.
Alkalis often feel soapy or slimy.
Alkalis can be corrosive and very damaging to skin.
Alkalis neutalise acids to make salts and water.
Alkalis dissolve in water to give an alkaline solution.
Alkalis are usually Metal Oxides, Hydroxides or Carbonates eg. sodium hydroxide (NaOH)

Last Updated on March 25, 2019 by Emma Vanstone





Chris Bernstein says
We actually do this at home. To avoid too much consumption of acid.
76Georgina says
Hi blogger, i must say you have high quality content here.
Your website should go viral. You need initial traffic
boost only. How to get it? Search for: Mertiso's tips go viral